 |
| Collection of samples in the region of Fukushima |
The accident at the Fukushima-1 released large quantities of radioactive material in the air: this is now dominated by radioactive isotopes of Cesium-137 and -134. In a previous post we compared measurements of environmental radioactivity, showing how the radiation background in Rome is higher than in Tokyo.
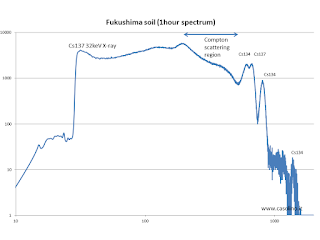
The figure shows the spectrum of gamma rays emitted by an artificial source of cesium-137 acquired with the same spectrometer described in the previous post.The cesium decays into an excited state of barium (emitting an electron and an antineutrino). The de-excitation of barium emits a gamma ray energy of 661 keV. This process is similar to fluorescence, except that this phenomenon is due to excited atomic electrons. The return of the electrons to the ground state emits light (between 2 and 3 eV), that is electromagnetic radiation. In fluorescence the energy levels in the nucleus are thousands of times more intense and therefore the electromagnetic radiation emitted has an energy thousands of times greater.
To the left (lower energy) of the 661 keV peak you have the " Compton shoulder ", produced by gamma rays hitting and producing an electron with lower energy than the primary gamma, depending on the angle with which it is issued.
On the far left there is also a peak at 30 keV is also due to the emission of X-ray excitation of barium.
The spectroscopic analysis of a sample of soil in the region of Fukushima collected in January 2013 shows a more complex structure. In this case it is a particularly contaminated sample, took the side of a mountain road between the city of Fukushima and the coast . Other samples are not so active. Presumably the deposition of water and snow did accumulate large amounts of cesium on the edge of the road. As already described in other posts, the eplosions at plant has emitted reactor also cesium-134 which decays into barium with peaks at 600, 790, 1400 and 1600 keV (the latter is out of range of the detector). Each gamna has its Compton shoulder associate: the sum of the various emission produces the characteristic spectrum shown in the figure.
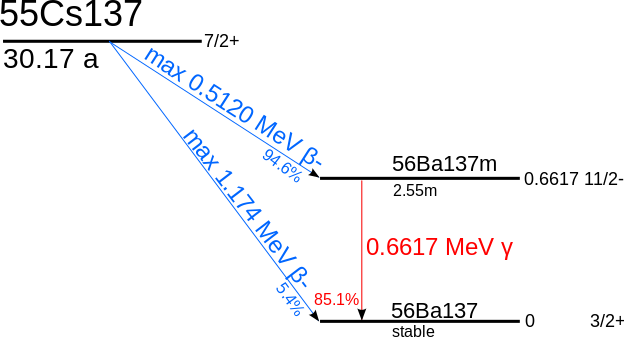 |
| Decay pattern of cesium-137 ( from here ) |
It should be recalled that the cesium-134 has a decay time of two years, for which the presence of these peaks denotes the origin of Fukushima. In other cases the absence of cesium-134 has been used to show how fungi which also had radioactivity above the threshold of 100 Bq / kg were not contaminated by the central, but presumably from previous nuclear tests.
From the decay probabilities and the height of the peaks can be traced to the relative abundances of different isotopes. Reconstructing the absolute activity of a given sample in Bq / kg is much more complicated because it is necessary to take into account the geometry and efficiency of the detector: it will be the subject of a future post.


Nessun commento:
Posta un commento