 The maximum amount of Cesium allowed in food produced in Japan has recently been lowered from 500 Bq/kg to 100 Bq/kg. This very conservative level is causing several problems to vegetable and meat producers, that have to throw away larger quantities of food.
The maximum amount of Cesium allowed in food produced in Japan has recently been lowered from 500 Bq/kg to 100 Bq/kg. This very conservative level is causing several problems to vegetable and meat producers, that have to throw away larger quantities of food.
Even though the value of bananas is 125 Bq/kg of potassium 40 and up to 260 Bq/kg of Radium in brazilian nuts is higher than the current limit, it is sometimes argued that the values are unrelated.
A conservative (and rough) estimate would give a factor 4 to "convert" from Cesium to Potassium (1Bq of) Cs = 4 Bq od K40.
In other words eating 100 g of Shitake with 50 Bq of Cesium is equivalent to eating a couple (200g) of bananas. This takes into account decay modes, physiology and time to expel it from the body. Calculations are below. Some points worth remembering are:
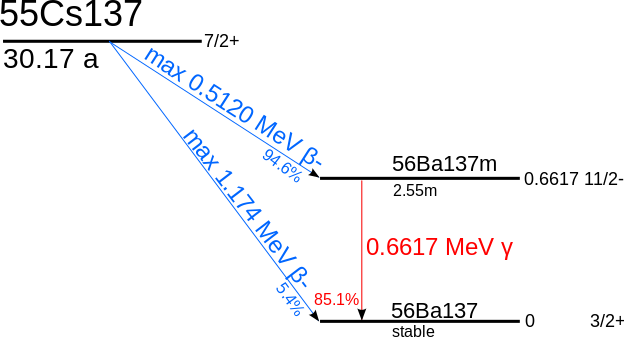 |
| Decay scheme of Cs137: either one electron (beta-) or one electron and one gamma. |
But radioactivity from bananas is "natural" and good! There is no "natural" (good) and "artificial" (bad) radioactivity. All radioactive elements decay in the same ways and are potentially dangerous. Ceisum and Potassium decay in the same way (electron or gamma) and there is no difference among the two.
Becquerels and Sieverts are two different things, to be used in different contexts. Becquerels represent the amount of radiation in an object (food, ground, trees...). Sieverts represent the equivalent dose to which the human body is exposed. Think of Becquerels as the luminosity of an UV light bulb of a tanning machine and Sieverts as the amount of tanning we might get from it. It is not straightforward to pass from Becquerels to Sieverts, since it depends on the exposure time, if the food is ingested etc...
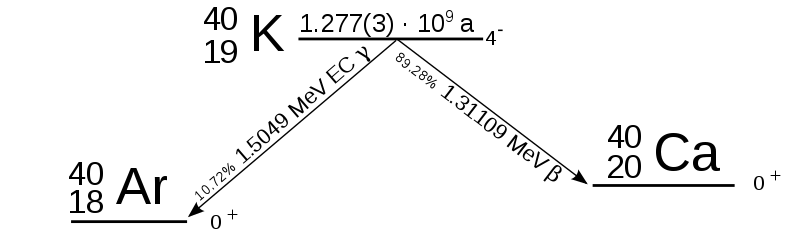 |
| Decay scheme of K40, either one gamma or one electron |
Does Cesium accumulate in human body? No, it is expelled in one to four months, compared to one month of Potassium:
Cesium 137 and 134. The biological half-life of Cesium is between one and four months. ICRP standard give 110 days for man and 65 days for women. Some quotations: (53+-12 days in children). 112 days in adults. Some differences between population may arise: biological half time is somewhat shorter among Scandinavian males (84 days), not in females (64 days).
Potassium 40. Our body contains about 3700 Bq of potassium 40, corresponding to about 0.32 mSv in an year. The biological half-life of potassium, that is the time it takes to expel half of it from our body is 30 days. (data from here).
So, how do I compare radiation from potassium and from Cesium?
On average, and very roughly, 100 Bq from Cesium results in 4 times more Sieverts than 100 Bq from Potassium. This takes into account the factor 2 of the decay (Cesium decays in 82% of the time in an electron and then one gamma) and another factor 2 for the biological half-life (two months for Cesium vs one month of Potassium). In the most conservative case you might assume a factor 8 (2 for the decay and 4 months of maximum half-life).
Nessun commento:
Posta un commento